Nuclear Physics and RadioactivityHard
Question
An electron in hydrogen atom first jumps from second existed state to first excited state and then, from first excited state to ground state. Let the ratio of wavelength, momentum and energy of photons in the two cases be x,y and z, then select the wrong answer /(s)
Options
A.z = 1/x
B.x = 9/4
C.y = 5/27
D.z = 5/27
Solution
E1 = 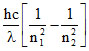
For second excited state to first excited state
E1 =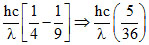
For first excited state to ground excited state
E2 =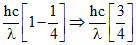
(A)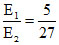 (B)
(B) 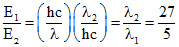
(C) P ∝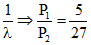
For second excited state to first excited state
E1 =
For first excited state to ground excited state
E2 =
(A)
(C) P ∝
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Consider a hydrogen like atom whose energy in nth excited state is given by En = - when this excited atom makes a transi...A radioactive element X converts into another stable element Y. Half-life of X is 2h. Initially, only X is present after...A radioactive element X disintegrates into a stable element Y with half life 2 year. A fresh sample of X is available at...The half life of a radioactive substance is 20 minutes. The approximate time interval (t2 - t1) between the time t2 when...A radioactive material decays by simultaneous emission of two particles with respective half−lives 1620 and 810 ye...