Nuclear Physics and RadioactivityHard
Question
The electron in a hydrogen atom jumps back from an excited state to ground state, by emitting a photon of wavelength λ0 = 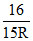 where R is Rydberg’s constant. In place of emitting one photon, the electron could come back to ground state by
where R is Rydberg’s constant. In place of emitting one photon, the electron could come back to ground state by
Options
A. Emitting 3 photons of wavelength λ1, λ2 and λ3 such that 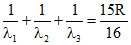
B. Emitting 2 photons of wavelength λ1, and λ2 such that 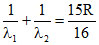
C. Emitting 2 photons of wavelength λ1, and λ2 such that λ1, + λ2 = 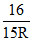
D. Emitting 3 photons of wavelength λ1, λ2 and λ3 such that λ1, + λ2 + λ3 = 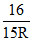
More Nuclear Physics and Radioactivity Questions
Which of the following pair of nuclei are isobars of the element?...A sample of radioactive material decays simultaneously by two processes A and B with half lives and hr respectively. For...If half-life of a radioactive substance is 60 minutes, then the percentage decay in 4 hours is :...When a hydrogen atom is exited from ground state to first excited state then :...Which statement about radioactive radiations is true...