Nuclear Physics and RadioactivityHard
Question
Which of the following transitions in hydrogen atoms emit photons of highest frequency ?
Options
A.n = 2 to n = 6
B.n = 6 to n = 2
C.n = 2 to n = 1
D.n = 1 to n = 2
Solution
∵ ᐃE = hv ∵ v ∝ 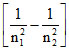
∴ vmax for n2 = 2 to n1 = 1
∴ vmax for n2 = 2 to n1 = 1
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
The photon radiated from hydrogen corresponding to 2nd line of Lyman series is absorbed by a hydrogen like atom ′X...The figure indicates energy level diagram of an atom and the origin of five spectral lines in emission spectra. Which of...A neutron collides head - on with a stationary hydrogen atom in ground state. Which of the following statements are corr...The order of magnitude of the density of a uranium nucleus is ($m_p = 1.67 \times 10^{-27}$ kg)...Light coming from a discharge tube filled with hydrogen falls on the cathode of the photoelectric cell. The work functio...