Nuclear Physics and RadioactivityHard
Question
The figure indicates energy level diagram of an atom and the origin of five spectral lines in emission spectra. Which of the spectral lines will also occur in the absorption spectra?
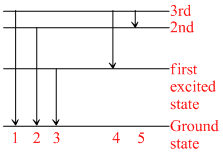

Options
A.1, 2, 4
B.2, 3, 4, 5
C.1, 2, 3
D.1, 2, 3, 4, 5
Solution
For absorpiton spectra, atom must be in ground state
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Force acting on proton-proton is -...The diagram shown the energy levels for an electron in a ace rain atom. Which transition shown represents the emission o...Which one moves with greatest speed :-...A radioactive sample disintegrates by 10% during one month. How much fraction will disintegrate in four months :-...A photoelecrtic cell illuminated by a point source of light 1 m away. When the source is Shifted to 2m then-...