JEE Advanced | 2011Mole ConceptHard
Question
A 5.2 molal aqueous solution of methyl alcohol, CH3OH , is supplied. What is the mole fraction of methyl alcohol in the solution ?
Options
A.0.190
B.0.086
C.0.050
D.0.100
Solution
Molefraction of solute (X2) in aqueous solution = 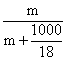
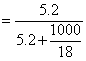
= 0.09
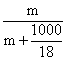
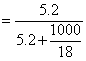
= 0.09
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Some bottles of colourless liquids were being labelled when the technicians accidentally mixed them up and lost track of...Two elements A and B combine to form compound X and Y. For the fixed mass of A, masses of B combined for the compounds X...To what extent must a given solution of concentration of 40 mg silver nitrate per ml be diluted to yield a solution of c...Silver metal in ore is dissolved by potassium cyanide solution in the presence of air by the reaction 4 Ag + 8 KCN + O2 ...The pair of species having identical shapes for molecules of both species is...