Nuclear Physics and RadioactivityHard
Question
A vessel of 831cc contains 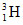 at 0.6 atm and 27oC. If half life of
at 0.6 atm and 27oC. If half life of 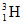 is 12.3 years then the activity of the gas is -
is 12.3 years then the activity of the gas is -
Options
A.3.04 × 1013 dps
B.582Ci
C.2.15 × 1012 dps
D.823 Ci
Solution
Number of moles of gas n = 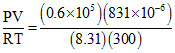 = 0.02
= 0.02
Activity = λN =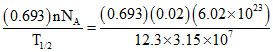 = 2.15 × 1013 dps
= 2.15 × 1013 dps
=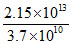 = 582 Ci
= 582 Ci
Activity = λN =
=
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
For a radioactive sample, at given instant, number of active nuclei is N and its decay constant is λ then the incor...The amount of radioactive substance left after one hour whose half life is 20 minutes is :...A radioactive sample consists of two distinct species having equal number of atoms initially. The mean life of one speci...Source of energy of the sun is :...What is ratio of volume of atom and nucleus :-...