ElectrochemistryHard
Question
The reduction potential of hydrogen half cell will be negative if :
Options
A.p(H2) = 1 atm and [H+ = 1.0 M
B.p(H2) = 2 atm and [H+ = 1.0 M
C.p(H2) = 2 atm and [H+ = 2.0 M
D.p(H2) = 1 atm and [H+ = 2.0 M
Solution
2H+ + 2e- → H2 (g)
Eo ″ 0.059 log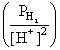 (here E is - ve when PH2 > [H+]2)
(here E is - ve when PH2 > [H+]2)
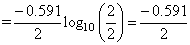 × 0.3010 = negative value
× 0.3010 = negative value
Eo ″ 0.059 log
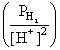 (here E is - ve when PH2 > [H+]2)
(here E is - ve when PH2 > [H+]2)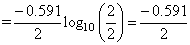 × 0.3010 = negative value
× 0.3010 = negative valueCreate a free account to view solution
View Solution FreeMore Electrochemistry Questions
The equilibrium Cu++ (aq) + Cu(s) ⇋ 2Cu+ established at 20oC corresponds to = 2.02 × 104+. The standard poten...In the electrochemical cell :- Zn|ZnSO4(0.01M)| |CuSO4(1.0 M)|Cu, the emf of this Daniel cell is E1. When the concentrat...Four successive members of the first row transition elements are listed below with atomic numbers. Which one of them is ...Using same quantity of current, which among Na, Mg and Al is deposited more (by mass) during electrolysis of their molte...Consider the following Eo values EoFe3+/Fe2+ = 0.77 V EoSn2+/Sn = - 0.14 V Under standard conditions the potential for t...