ElectrochemistryHard
Question
The equilibrium Cu++ (aq) + Cu(s) ⇋ 2Cu+ established at 20oC corresponds to 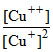 = 2.02 × 104+. The standard potential, EoCu++,Cu = 0.33 volt at this temperature. What is the standard potential, EoCu++, Cu ?
= 2.02 × 104+. The standard potential, EoCu++,Cu = 0.33 volt at this temperature. What is the standard potential, EoCu++, Cu ?
Options
A.- 0.457 V
B.- 0.125 V
C.- 0.66 V
D.- 0.250 V
Solution
At equilibrium
Eo1 = +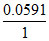 log10
log10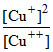
=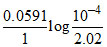 = - 0.254
= - 0.254
EoCu2+/Cu+ - EoCu+/Cu
and EoCu+2/Cu =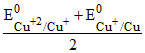
so Eo1 = 2EoCu+2/Cu = - 2EoCu+2/Cu
EoCu+/Cu = 0.457
EoCu/Cu+ = - 0.457
Eo1 = +
=
EoCu2+/Cu+ - EoCu+/Cu
and EoCu+2/Cu =
so Eo1 = 2EoCu+2/Cu = - 2EoCu+2/Cu
EoCu+/Cu = 0.457
EoCu/Cu+ = - 0.457
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A cell whose resistance, when filled with 0.1 M – KCl is 200 Ω, is measured to be 6400 Ω, when filled with 0.003 M – NaC...Select the correct option if it is known that Ksp (AgCl) > Ksp (AgBr) > Ksp (AgI)...The specific conductance of a N/10 KCl at 25oC is 0.0112 ohm-1 cm-1. The resistance of cell containing solution at the s...A current is passed through 2 voltameters connected in series. The first voltameter contains XSO4 (aq.) and second has Y...The current efficiency of an electrode position of copper metal in which 9.8 g of copper is deposited by a current of 3 ...