Heat and Thermal ExpansionHard
Question
5g of steam at 100oC is mixed at 100oC is mixed with 10g of ice at 0oC. Chose correct alternative/s) :- (Given swater = 1 cal / goC, LF = 80 cal / g, LV = 540 cal / g)
Options
A.Equilibrium ,temperature of mixture is is 160oC
B.Equilibrium ,temperature of mixture is is 100oC
C.At equilibrium, mixture contain 13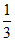 g of water
g of water
D.At equilibrium, mixture contain 1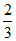 g of steam
g of steam
Solution
Required heat Available heat
10 g ice (0oC) 5 g steam (100oC)
↓800 cal ↓ 2700 cal
10 g water (0oC) 5 g water (100oC)
↓1000 cal
10 g water (100o)
So available heat is more than required heat therefore final temperature will be 100oC.
Mass of vapour condensed
=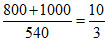 g
g
Total mass of water
= 10 +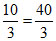 = 13
= 13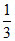 g
g
Total mass of steam
= 5 -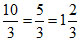 g
g
10 g ice (0oC) 5 g steam (100oC)
↓800 cal ↓ 2700 cal
10 g water (0oC) 5 g water (100oC)
↓1000 cal
10 g water (100o)
So available heat is more than required heat therefore final temperature will be 100oC.
Mass of vapour condensed
=
Total mass of water
= 10 +
Total mass of steam
= 5 -
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Two conductors have the same resistance at 0oC but their temperature coefficients of resistance are α1 and α2....Given T - P curve for three processes. If initial and final pressure are same for all processes than work done in in pro...A thermodynamic system undergoes cyclic process ABCDA as shown figure. The work done by the system is : -...One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A...A fine wire of length 4m is fixed rigidly in a heavy in a brass frame as shown in figure. It is just taut at 20oC. The t...