Ionic EquilibriumHard
Question
Some chemists at ISRO wished to prepare a saturated solution of a silver compound and they wanted it to have the highest concentration of silver ion possible. Which of the following compounds would they use ?
Given:-
Ksp (AgCl) = 1.8 × 10-10.
Ksp (AgCl) = 1.8 × 10-10.
Ksp(AgBr) = 5.0 × 10-13,
Ksp (Ag2CrO4) = 2.4 ×10-12 [use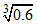 = 0.84]
= 0.84]
Ksp (Ag2CrO4) = 2.4 ×10-12 [use
Options
A.AgCl
B.AgBr
C.Ag2CrO4
D.All of these
Solution
Find the [Ag+] in all the saturated solutions.
[Ag+]in AgCl =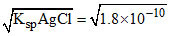 M;
M;
[Ag+]in AgBr =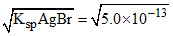 M
M
[Ag+]in AgrCrO4 = 2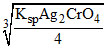
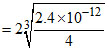
⇒ [Ag+] is maximum in saturated solution of Ag2CrO4.
[Ag+]in AgCl =
[Ag+]in AgBr =
[Ag+]in AgrCrO4 = 2
⇒ [Ag+] is maximum in saturated solution of Ag2CrO4.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A certain indicator (an organic dye) has pKa = 5. For which of the following titrations may it be suitable....Which of the following salts undergoes anionic hydrolysis?...How many grams of HCl should be dissolved in sufficient water to get 500 ml of an aqueous solution of pH, 2.0?...To 0.35 L of 0.1 M – NH3 0.15 L of 0.1 M-MgCl2 is added. What minimum mass of (NH4)2SO4 should be added to cause the Mg(...The sodium salt of a certain weak monobasic organic acid is hydrolysed to an extent of 3% in its 0.1M solution at 25oC. ...