Hydrogen and its compoundHard
Question
The pKb of CN- is 4.7. The pH of solution prepared by mixing 2.5 mol of KCN of 2.5 mol of HCN in water and making the total volume upto 500 mL is
Options
A.10.3
B.~ 2.4
C.~2.8
D.~ 1.6
Solution
It forms basic buffer
pOH = pKb + log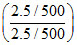 = 4.7
= 4.7
pH = 14 - 4.7 = 9.3
pOH = pKb + log
pH = 14 - 4.7 = 9.3
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
Which compound does not dissolve in hot, dilute HNO3 ?...Concentrated hydrochloric acid when kept in open air sometimes produces a cloud of white fumes. The explanation for it i...Which of the following transition will emit maximum energy in hydrogen atom :-...The geometry of Ni(CO)4 and Ni(PPh3)2 Cl2 are :...In hydrogen spectrum, If n = 4 then ratio of number of lines in emission spectrum, in the ultra violet and visible regio...