Hydrogen and its compoundHard
Question
The geometry of Ni(CO)4 and Ni(PPh3)2 Cl2 are :
Options
A.both square planar
B.tetrahedral and square planar, respectively
C.both tetrahedral
D.square planar and tetrahedral, respectively
Solution
In both the complexes central Ni-atom is sp3 -hybridised and both are tetrahedral. Their structures are as given below :
Ni(CO)4 : Ni(0):
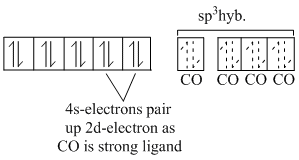
[Ni(PPh3)2] : Ni (II):
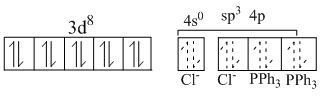
because bothe Cl- and PPh3 are weak ligands and so the d-electron are not paired to make 3d-orbital available for hybridization. Again 4sp3h. Os. take part bonding and geometry is tetrahedral.
Ni(CO)4 : Ni(0):

[Ni(PPh3)2] : Ni (II):

because bothe Cl- and PPh3 are weak ligands and so the d-electron are not paired to make 3d-orbital available for hybridization. Again 4sp3h. Os. take part bonding and geometry is tetrahedral.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
Which of the following transition will emit maximum energy in hydrogen atom :-...One of the constituent of German silver is :...Ammonium dichromate is some fireworks. The green coloured powder blown in the air is :...The number of σ and π bonds present in ethane is :...Identify alkene which has maximum heat of hydrogenation ?...