Heat and Thermal ExpansionHard
Question
The specific heats of a gas are CP = 0.2 cal / g oC & CV = 0.15 cal / g oC .[Take R = 2 cal / mol oC]
Options
A.The molar mass of the mass of the gas is 40g
B.The molar mass of the gas cannot be determined from the data given
C.The number of degree of freedom of the gas molecules is 6
D.The number of degree of freedom of the gas molecules is 8
Solution
CP - CV = R; M (0.20 - 0.15) = 2 [M = molar mass]
⇒ M =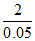 = 40g
= 40g
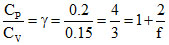
⇒ f = degrees of freedom = 6
⇒ M =
⇒ f = degrees of freedom = 6
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A...Density of water at $4^{\circ}C$ and $20^{\circ}C$ are $1000\text{ }kg/m^{3}$ and respectively. The increase in internal...The figure shows two rods, one made of aluminum [α = 23 × 10-6 (Co)-1] and the other of steel [α = 12 ...Four moles of hydrogen , teo moles of helium and one mole of water vapor from an ideal gas mixture. What is the molar sp...On an X temperature scale, water freezes at - 125.0oX and boils at 375.0oX On a Y temperature scale, water freezes at - ...