Heat and Thermal ExpansionHard
Question
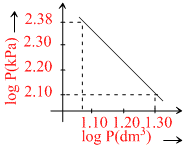
Logarithms of readings of pressure and volume fo ran ideal gas plotted on a graph as shown in figure. By measuring the gradient, It can be shown that the gas may be : -

Options
A.Monoatomic and undergoing an adiabatic change.
B.Monotomic and undergoing an isothermal change
C.Diatomic and undergoing an adiabatic change
D.Triatomic and undergoing an isothermal change
Solution
PVγ = C; ln P + γlnV = lnC
⇒ lnP = - γlnV = lnC ⇒ Y = mx + c
m = - γ = -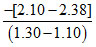 = - 1.4
= - 1.4
∴ The gas is diatomic
⇒ lnP = - γlnV = lnC ⇒ Y = mx + c
m = - γ = -
∴ The gas is diatomic
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A inert gas obeys the law PVx = constant. For what value of x, it has negative molar specific heat -...Air is filled at 60oC in s vessel of open mouth. The vessel is heat to a temperature T so that part of air escapes. The ...If S is stress and Y is Young′s modulus of material of a wire, the energy stored in the wire per unit volume is...The temperature of the two outer surface of a composites slab, consisting of two materials having coefficients of therma...A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S into two equal parts A and B. ...