Heat and Thermal ExpansionHard
Question
A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S into two equal parts A and B. Part A has an Ideal gas at pressure P0 and temperature T0 and in part B is vacuum. A mass less spring of force constant k is connected with piston and the wall of the container as shown. Initially spring is unstructured. Gas in chamber A is allowed to expand. Let in equilibrium spring is compressed by x0. Then : -
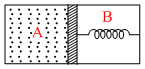

Options
A.Final pressure of the gas is 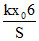
B.Work done by the gas is 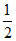 kx20
kx20
C.Change in internal energy of the gas is 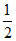
D.Temperature of the gas is decreased.
Solution
Final pressure 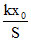
Workdone by gas = p.E. stored in spring = 1/2 kx02
ᐃU = |-ᐃW| = 1/2 kx02
As gas expands, ᐃT is negative.
Workdone by gas = p.E. stored in spring = 1/2 kx02
ᐃU = |-ᐃW| = 1/2 kx02
As gas expands, ᐃT is negative.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A point source of heat of power P is placed at the center of a spherical shell of mean radius R the material of the shel...Two cylinders A and B fitted with piston contain the equal amount of an ideal diatomic gas at 300K. The piston of A is f...N(< 100) Molecules of a gas have velocities 1, 2, 3.........N , km/s respectively . Then :-...The temperature of an isotropic cubical solid of length l0 density ρo and coefficient of liner expansion is increas...One end of a horizontal thick copper wire of length 2L and radius 2R is welded to an end of another horizontal thin copp...