Heat and Thermal ExpansionHard
Question
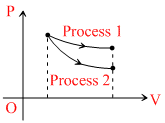
The indicator diagram for two process 1 and 2 carried on an ideal gas is shown in figure. If m1 and m2 be the 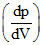 for process 1 and process 2 respectively, them :-
for process 1 and process 2 respectively, them :-

Options
A.m1 = m2
B.m1 > m2
C.m1 < m2
D.m2CV = m1CP
Solution
Slope of isothermal process
m1 =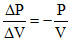
Slope of adiabatic process
m2 =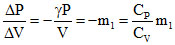
m1 =
Slope of adiabatic process
m2 =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
5n, and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each ot...A thermocouple of negligible resistance produces an e.m.f of 40 μ V/oC in the linear range of temperature. A galvan...A gas ha volume V and persuure P. total translational kinetik energy of all the molecules of the gas is :-...Twelve conditioning rods from the sides of a uniform cube of side l. If in steady state, B and H ends of the cube are 10...A glass rod measured with a zinc scale, both being at 30oC appears to be of length 100cm. If the scale shows correct rea...