Heat and Thermal ExpansionHard
Question
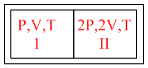
A partition divides a container having insulated in to two compartments I and II. The same gas fills the two compartments whose initial parameter are given. The partition is a a conducting wall which can move freely with out friction. Which of the following statement is / are correct, with reference to the final equilibrium position ?

Options
A.The pressure in the two compartments are equal.
B.Volume of compartment I is 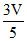
C.Volume of compartment it is 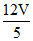
D.Final pressure in compartment I is 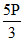
Solution
For insulated chambers n1 + n2 = n1′ + n2′
(final pressures becomes equal)
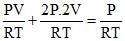 [3V] ⇒ P′ =
[3V] ⇒ P′ = 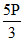
For left chamber
PV = PV′ =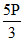 V ; ⇒ V′ =
V ; ⇒ V′ = 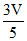
For right chamber
4PV = PV′ =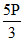 ⇒ V′ =
⇒ V′ = 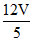
(final pressures becomes equal)
For left chamber
PV = PV′ =
For right chamber
4PV = PV′ =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A metal rod A of length by ᐃl when its temperature is raised by 100oC. Another rod B of different metal of length ...The internal energy of as gas is given U = 5 + 2PV. It expands from V0 to 2V0 against a constant pressure P0. The heat a...In a 10-meter-deep lake, the bottom is at a constant temperature of 4oC. The air temperature is constant at - 4oC. the t...One mole of an ideal gas undergoes P = Here P0 and V0 constant. Change in temperature of the gas when volume is changed ...Consider the MB shown in the diagram, let the resistance X have temperature coefficient and the resistance from the RB h...