Heat and Thermal ExpansionHard
Question
One mole of an ideal gas undergoes P = 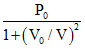 Here P0 and V0 constant. Change in temperature of the gas when volume is changed from V = V0 to V = 2V0 is :-
Here P0 and V0 constant. Change in temperature of the gas when volume is changed from V = V0 to V = 2V0 is :-
Options
A.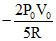
B.
C.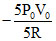
D.P0V0
Solution
Ideal gas equation PV nRT
So at V =V0; RT =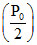 (V0) and at V = 2V0,
(V0) and at V = 2V0,
RT2 =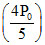 (2V0) ⇒ T2 - T1 =
(2V0) ⇒ T2 - T1 = 
So at V =V0; RT =
RT2 =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
On an X temperature scale, water freezes at - 125.0oX and boils at 375.0oX On a Y temperature scale, water freezes at - ...Two fine steel wires fastened between the projections of a heavy brass bar,are just taut when the whole system is at 0oC...5n, and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each ot...Three rod of some dimensions are arranged as shown in the figure. They have thermal conductivities k1, k2 & k3. The poin...An vessel conatins 14 g (moles) of hydrogen and 96 g (3 moles) of oxygen at ATP. Chemical reaction induced by passing el...