Heat and Thermal ExpansionHard
Question
A gas undergoes an adiabatic process in which pressure becomes 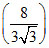 times and volume become
times and volume become 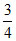 of initial volume. If initial absolute temperature was T, the final temperature is
of initial volume. If initial absolute temperature was T, the final temperature is
Options
A.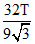
B.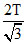
C.T3/2
D.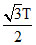
Solution
For adiabatic process, P1V1γ = P2V2γ ⇒ P0V0γ = 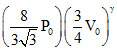 ...(i)
...(i)
Also, T1V1γ-1; = T2V2γ-1; ⇒ TV0γ-1 = T2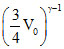 ...(ii) Solving (i) & (ii), T2 =
...(ii) Solving (i) & (ii), T2 = 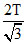
Also, T1V1γ-1; = T2V2γ-1; ⇒ TV0γ-1 = T2
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The pressure that has to be applied to the ends of a steel rod of length 10 cm to keep its length constant when its temp...Ice starts forming in like with water at 0oC when the temperature is - 10oC. If the taken for 1 cm of ice be 7 hours, th...On mixing 10 mL of carbon tetrachloride with 10 mL of benzene, the total volume of the solution is...Three identical adiabatic containers have helium, neon and oxygen gases at the same pressure.The gases are compressed to...Radius of a conductor increase informally from left end to right end as shown in Fig. Material of the conductor is isotr...