Heat and Thermal ExpansionHard
Question
The internal energy of a gas in an adiabatic process is given by U = a + bPV, find γ : -
Options
A.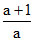
B.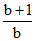
C.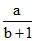
D.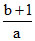
Solution
U = a + bPV = a + bnRT
⇒ ᐃU = bnRᐃT = nCvᐃT
⇒ Cv = bR ⇒ Cp = bR +R
⇒ γ =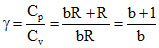
⇒ ᐃU = bnRᐃT = nCvᐃT
⇒ Cv = bR ⇒ Cp = bR +R
⇒ γ =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A meter washer has a hole of diameter d1 and external d2, where d2 = 3d1. On heating, d2 increase by 0.3%. Then d1 will ...Three long straight wires carrying current are arranged mutually parallel as shown in the figure. The force experienced ...The amount of work done in stretching a spring from a stretched length of 10 cm to a stretched length of 20 cm is-...An vessel conatins 14 g (moles) of hydrogen and 96 g (3 moles) of oxygen at ATP. Chemical reaction induced by passing el...The emissive of a black body at T = 300 K is 100 Watt/m2. Consider a body B of area A = 10 m2, coefficient of reflectivi...