Heat and Thermal ExpansionHard
Question
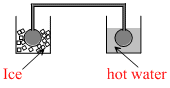
Two identical glass bulbs are interconnected by a thin glass tube at 0oC A gas is filled at N.T.P in these bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes 1.5 The temperature of hot bath will be : -

Options
A.100oC
B.180oC
C.256oC
D.546oC
Solution
Number of moles remain constant
n1 + n2 = n1′ + n2′ ⇒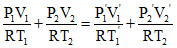
⇒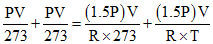
⇒ T = 273 × 3 k = (273 × 3 - 273)oC = 546oC
n1 + n2 = n1′ + n2′ ⇒
⇒
⇒ T = 273 × 3 k = (273 × 3 - 273)oC = 546oC
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
In a mercy = glass thermometer the cross - section of the capillary portion is A0 and the volume of the bulb is V0 at 27...A thermodynamic system undergoes cyclic process ABCDA as shown figure. The work done by the system is : -...At 4oC, 0.98 of the volume of a body in immersed in water. The temperature at which the entire body gets immersed in wat...Hailstone at 0oC falls from a height of 1 km on an insulating surface converting whole of its kinetic energy into heat. ...Suppose 0.5 mole of an ideal gas undergoes an isothermal expansion as energy is added to it as heat Q. Graph shows the f...