Heat and Thermal ExpansionHard
Question
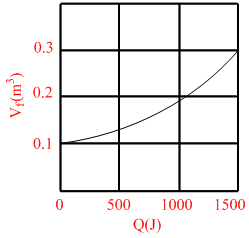
Suppose 0.5 mole of an ideal gas undergoes an isothermal expansion as energy is added to it as heat Q. Graph shows the final volume Vf versus Q. temperature of the gas is :- (use ln 9 = 2 and R = 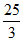 J/ mole - K)
J/ mole - K)

Options
A.293 K
B.360 K
C.386 K
D.412 k
More Heat and Thermal Expansion Questions
A fine wire of length 4m is fixed rigidly in a heavy in a brass frame as shown in figure. It is just taut at 20oC. The t...540 g of ice at 0oC is mixed with 540g of water at 80oC. The temperature of the mixture is (Give latent heat of fusion o...Pressure versus temperature graph of an ideal gas is shown in figure. Density of the gas at point A is. ρ0 Density ...A spherical body with an initial temperature T1 is allowed to coll in surrounding at temperature T0(< T1). The mass o...A gas ha volume V and persuure P. total translational kinetik energy of all the molecules of the gas is :-...