Chemical BondingHard
Question
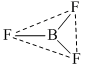
The molecule which has zero dipole moment is :
Options
A.CH2Cl2
B.BF3
C.NF3
D.ClO2
Solution
due to plamar symmetrical structure μR = 0

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In the given reaction C6H5 - O - CH2 - CH3 [X] + [Y] [X] and[Y] will respectively be :-...Dipole induced dipole interactions are present in which of the following pairs :-.......................(1) ....................(2)If both the reaction shows kinetic isotope effect then...Determine C-C & C-H bond enthalpy (in kJ/mol)Given ᐃfHo(C2H6, g) = - 85 kJ/molᐃfHo(C3H8, g) = - 104 kJ mol&#...Which of the following compound has a P-P bond ?...