Heat and Thermal ExpansionHard
Question
Pressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement :-
(i)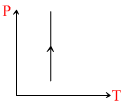 (ii)
(ii) 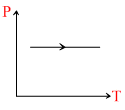 (iii)
(iii) 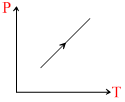
(i)
 (ii)
(ii)  (iii)
(iii) 
Options
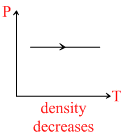
A.Density of gas is increasing in graph (i)
B.Density of gas decreasing in graph (ii)
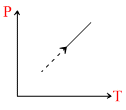
C.Dencity of gas is constant in graph (iii)
D.None of these
Solution
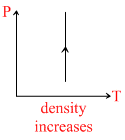
For equation :
At constant temperature
For 1st graph : ρ ∝ P At constant temperature.
For 2nd graph :
For equation :
At constant P, ρ ∝
For 3rd graph L. :
⇒ density = ρ constant
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A block body emits at the rate P when its temperature is T. At this temperature the wavelength at which the radiation ha...Suppose 0.5 mole of an ideal gas undergoes an isothermal expansion as energy as added it as heat Q. Graph shows the fina...A triangular plate has two cavities one square and one regular as shown figure The plate is heated....In a 20 m deep lake, the bottom is at a constant temperature of 4oC. the air temperature is constant at -10oC. The therm...5g of steam at 100oC is mixed at 100oC is mixed with 10g of ice at 0oC. Chose correct alternative/s) :- (Given swater = ...