Chemical Kinetics and Nuclear ChemistryHard
Question
The reaction A(g) + 2B(g) → C(g) + D(g) is an elementary process. In an experiment, the initial partial pressure of A and B PA = 0.60 and PB = 0.80 atm. when PC = 0.2 atm, the rate of reaction relative to the initial rate is
Options
A.1/48
B.1/24
C.9/16
D.1/6
Solution
r1 = k[A][B]2 = k[0.6] [0.80]2
After reaction
A + 2B → C + D
0.06 - 0.2 0.8 - 0.4 0.2 0.2
0.4 0.4 0.2 0.2
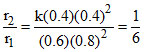
After reaction
A + 2B → C + D
0.06 - 0.2 0.8 - 0.4 0.2 0.2
0.4 0.4 0.2 0.2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Atomic number of a radioactive element is 100. It first decays into an element Y which then decays into Z. In both the p...The decomposition of N2O5 according to the equation 2N2O5(g) → 4NO2(g) + O2(g) is a first order reaction. After 30 minut...The compound used in enrichment of uranium for nuclear power plant is :...For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation...In a study of effect of temperature on reaction rate, the value of $\frac{1}{K}.\frac{dK}{dT}$ is found to be $\frac{1.2...