Ionic EquilibriumHard
Question
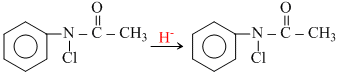
The reaction kinetics can be studied by
Options
A.Measurement of pH
B.Titration with hypo after adding KI
C.Both correct
D.None is correct
Solution
Cl attached to N in reactant oxidizes KI to I2 which can be determined by hypo (Na2S2O3).
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of following can act as buffer ?...For a weak electrolyte (HA) dissociation as lim c → 0, then:...The solubility of AgCl in water is 0.001435 g per litre at 15o C. The solubility product of AgCl is(Ag = 108, Cl = 35.3)...Consider a titration of potassium dichromate solution with acidified Mohr′s salt solution using diphenylamine as i...A volume of 2.5 ml of 2/5 M weak monoacidic base (Kb = 1 × 10−12 at 25o C) is titrated with (2/15) M-HCl in water at 25o...