Chemical BondingHard
Question
The rate expression for the reaction:
NH4CNO ⇋ NH2CONH2 can be derived from he mechanism:
i. NH4CNO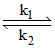 NH2NCO (Fast)
NH2NCO (Fast)
ii. NH4NCO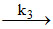 NH3 + HNCO (Fast)
NH3 + HNCO (Fast)
iii. NH3 + HNCO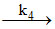 NH2CONH2 (Slow)
NH2CONH2 (Slow)
Which of the following statement(s) is/are correct about the rate expression ?
NH4CNO ⇋ NH2CONH2 can be derived from he mechanism:
i. NH4CNO
ii. NH4NCO
iii. NH3 + HNCO
Which of the following statement(s) is/are correct about the rate expression ?
Options
A.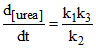 [NH4NCO]
[NH4NCO]
B.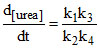 [NH4NCO]
[NH4NCO]
C.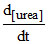 = k[NH4NCO]
= k[NH4NCO]
D.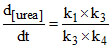 [NH4NCO]
[NH4NCO]
Solution
Applying steady state approximation to HNCO or NH3
∴
Also, [NH4NCO] =
∴
= k[NH4CNO]
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The maximum radius of sphere that can be fitted in the octahedral hole of cubical closed packing of sphere of radius r i...Which of the following comound is covalent ?...Bond order of 1.5 is shown by:...Consider the following species: CN+, CN–, NO and CN Which one of these will have the highest bond order?...Which of the following is correct :-...