Chemical BondingHard
Question
Bond order of 1.5 is shown by:
Options
A.O2
B.O2+
C.O2-
D.O22-
Solution
Bond Order = 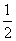 (Nb - Na) =
(Nb - Na) = 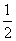 )10 - 7) = 1.5
)10 - 7) = 1.5
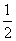 (Nb - Na) =
(Nb - Na) = 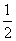 )10 - 7) = 1.5
)10 - 7) = 1.5Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following molecules has the maximum dipole moment?...Which is not correct order of given property :...Correct increasing order for the wavelengths of absorption in the visible region the complexes of Co3+ is :-...Element X is strongly electropositive and element Y is strongly electrongative. Both are univalent. The compound formed ...The product formed in the reaction of SOCl2 with white phosphorous is...