ElectrochemistryHard
Question
The Ecell for a given cell is 1.2346 and 1.2340 V at 300 K and 310 K, respectively. Calculate the change in entropy during the cell reaction if the redox change involves three electrons
Options
A.-17.37 J K-1
B.+17.37 J K-1
C.173.7 J K-1
D.5.79 J K-1
Solution
By ᐃS = 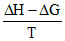
ᐃH = - nF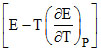 and ᐃG = - nEF
and ᐃG = - nEF
ᐃS = - nF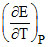 = 3 × 96500 ×
= 3 × 96500 × 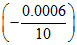
= - 17.37 JK-1
ᐃH = - nF
ᐃS = - nF
= - 17.37 JK-1
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to...2Ce4+ + Co → 2Ce3+ + Co2+ Eocell = 1.89 V, EoCo2+/Co = - 0.277 V hence, EoCe4+Ce3+ is :...The value of molar conductivity of HCl is greater than that of NaCl at a particular temperature because...Although aluminium is above hydrogen in the electrochemical series, it is stable in air and water. Why?...How many electrons flow when a current of 5 A is passed through a solution for 200 s?...