ElectrochemistryHard
Question
The resistance of 0.1 N solution of formic acid is 200 ohm and cell constant is 2.0 cm-1. The equivalent conductivity (in S cm2. eq-1) of 0.1N formic acid is :-
Options
A.100
B.10
C.1
D.None of these
Solution
k = G.G* ⇒ 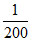 × 2 ⇒ 0.01;
× 2 ⇒ 0.01;
Λeq =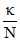 × 1000
× 1000
⇒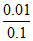 × 1000 ⇒ 100
× 1000 ⇒ 100
Λeq =
⇒
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The resistance of 1 M – CH3COOH solution is 250 Ω, when measured in a cell of cell constant 125 m−1. The molar conductiv...Which of the following is/are function(s) of salt - bridge ?...A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratio’s x:y and y:x has electrode potent...The electrode potential becomes equal to standard electrode potential when reactants and products concentration ratio is...The standard reduction potentials for Zn2+ / Zn,  Ni2+ / Ni and Fe2+ / Fe are - 0.76, - 0.23 and - 0.44 V respectiv...