Solid StateHard
Question
What is the maximum number of layers of atoms in close packed planes that will lie within two imaginary parallel planes having a distance between them of 13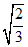 (where r is the radius of atoms) in the copper crystal (fcc)?
(where r is the radius of atoms) in the copper crystal (fcc)?
(Consider the atoms to be within the parallel planes if their enters are on or with in the two parallel planes).
(Consider the atoms to be within the parallel planes if their enters are on or with in the two parallel planes).
Options
A.5
B.6
C.7
D.8
Solution
Distance between two layer (A and B) = 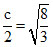 (r)
(r)
=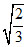 (2r)
(2r)
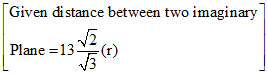
Let K is the number of imaginary planes.
Hence.
K ×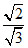 (2r = 13 )
(2r = 13 )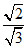 (r) ⇒ K =
(r) ⇒ K = 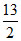 = 6.5 = 7
= 6.5 = 7
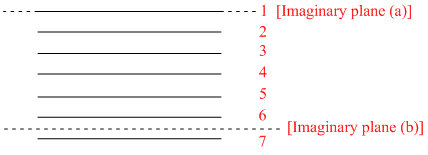
Thus, maximum number of layers = 7 (sec figure below).
=
Let K is the number of imaginary planes.
Hence.
K ×
Thus, maximum number of layers = 7 (sec figure below).

Create a free account to view solution
View Solution FreeMore Solid State Questions
The edge length of a face centered cubic cell of an ionic substance (NaCl) is 508 pm. If the radius of the cation is 110...Which one of the following schemes of ordering closed packed sheets of equal sized spheres does not generate close packe...Which of the following defects, if present, lowers the density of the crystal?...If the volume occupied by a unit cell of NaCl is 4.7 × 10–23 ml, then the volume of a crystal of NaCl weighing 1.0 g is...An ionic solid is HCP of Q2− ions and Px+ ions are in half of the tetrahedral voids. The value of x should be...