Coordination CompoundHard
Question
Which one of the following cyano complexes would exhibit lowest paramagnetic behaviour?
Options
A.[Cr(CN)6]3-
B.[Co(CN)6]3-
C.[Fe(CN)6]3-
D.[Mn(CN)6]3-
Solution
Co3+, E.C. = [Ar]3d6
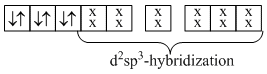
No, unpaired electron is present. Hence, paramagnetism in this complex is zero.
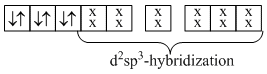
No, unpaired electron is present. Hence, paramagnetism in this complex is zero.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Geometrical shapes of the complexes formed by the reaction of Ni2+ with Cl-, CN- and H2O, respectively, are...The total number of possible coordination isomer for the given compound is[Pt(NH3)4Cl2][PtCl4]....Which of the following statements is correct with respect to the metal carbonyls of Ist transition series?...Which one of the following cyano complexes would exhibit the lowest value of paramagnetic behaviour ? (At. No. Cr = 24, ...Select the correct statement from the following ....