ElectrochemistryHard
Question
What is the standard potential of the Tl3+/Tl electrode ? if Eo Tl3+/Tl+ = 1.26 V, EoTl+/Tl+ = - 0.336 V
Options
A.0.728 V
B.0.118 V
C.1.728 V
D.2.728 V
Solution
Tl3+ + 2e- → Tl+; nFEo = 2 × 1.26 × F = 2.52F
Tl+ + e+ → Tl; nFEo = 1 ×(-0.336) × F = - 0.336F
Adding
Tl3+ + 3e- → Tl Eo =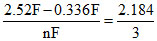
= 0.728 V
Tl+ + e+ → Tl; nFEo = 1 ×(-0.336) × F = - 0.336F
Adding
Tl3+ + 3e- → Tl Eo =
= 0.728 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Two electrochemical cells are assembled in which the following reactions occur:V2+ + VO2+ + 2H+ → 2V3+ + H2O; E°Cell = 0...Which one of the following statements is incorrect regarding an electrochemical cell?...If the cell reaction is spontaneous then...When the electric current is passed through a cell having an electrolyte, the positive ions move towards cathode and neg...The EMF of cell: H2(g)|Buffer||Normal calomel electrode, is 0.70 V at 25°C, when barometric pressure is 760 mm. What is ...