Hydrogen and its compoundHard
Question
26.8 gm of Na2SO4.nH2O contains 12.6 gm of water. The value of ′n′ is ?
Options
A.1
B.10
C.6
D.7
Solution
Na2SO4.nH2O
Molar mass = (142 + 18n)
Mass of water =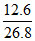 × (142 + 18n)
× (142 + 18n)
18n =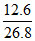 × (142 + 18n)
× (142 + 18n)
n = 7
Molar mass = (142 + 18n)
Mass of water =
18n =
n = 7
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
The number of σ and π bonds present in ethane is :...Ammonium dichromate is some fireworks. The green coloured powder blown in the air is :...″Metals are usually not found as nitrates in their ores″. Out of the following two (a and b) reasons which i...The enthalpy of combustion at 25oC of H2, cyclohexane and cyclohexene are - 241, - 3920 and - 3800 kJ/mol respectively. ...3-Ohenylpropene on reaction with HBr give (as)a major product):...