Hydrogen and its compoundHard
Question
The enthalpy of combustion at 25oC of H2, cyclohexane and cyclohexene are - 241, - 3920 and - 3800 kJ/mol respectively. The heat of hydrogenation of cyclohexene is?
Options
A.-121 kJ/mol
B.+ 121 kJ/mol
C.-242 kJ/mol
D.+ 242 kJ/mol
Solution
(i) H(g) + 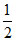 O2(g) → H2O(l) ᐃH = - 241 kJ
O2(g) → H2O(l) ᐃH = - 241 kJ
(ii) C6H10 +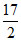 O2(g) → 6CO2(g) + 5H2O(l) ᐃH =- 3800 kJ
O2(g) → 6CO2(g) + 5H2O(l) ᐃH =- 3800 kJ
(iii) C6H12 + 9O2(g) → 6CO2(g) + 6H2O(l) ᐃH = - 3920 kJ
(i) + (ii) - (iii) give
ᐃH = - 241 - 3800 (- 3920) = - 121 kJ
(ii) C6H10 +
(iii) C6H12 + 9O2(g) → 6CO2(g) + 6H2O(l) ᐃH = - 3920 kJ
(i) + (ii) - (iii) give
ᐃH = - 241 - 3800 (- 3920) = - 121 kJ
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
Hydrogen peroxide oxidises [Fe(CN)6]4- to [Fe(CN)6]3- in acidic medium but reduces [Fe(CN)6]3- to [Fe(CN)6]4- in alkalin...Iron is rendered passive by treatment with concentrated :...Given :-The enthalpy of the hydrogenation of these compounds will be in the order as :-...Among the following insecticide is :...In which case is the number of molecules of water maximum?...