ThermochemistryHard
Question
Enthalpy change for the reaction,
4H(g) → 2H2 (g) is - 869.6 kJ
The dissociation energy of H - H bond is
4H(g) → 2H2 (g) is - 869.6 kJ
The dissociation energy of H - H bond is
Options
A.- 869.6 kJ
B.+ 434.8 kJ
C.+ 217.4 kJ
D.- 434.8 kJ
Solution
4H(g) → 2H2 (g) ; ᐃH = - 869.6 kJ
2H2(g) → 4H(g) ; ᐃH = 869.6 kJ
H2(g) → 2H(g) ; ᐃH =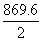 = 434.8 kJ
= 434.8 kJ
2H2(g) → 4H(g) ; ᐃH = 869.6 kJ
H2(g) → 2H(g) ; ᐃH =
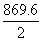 = 434.8 kJ
= 434.8 kJCreate a free account to view solution
View Solution FreeMore Thermochemistry Questions
How much energy is released when 6 moles of octane is burnt in air ? Given ᐃHfo for CO2(g), H2O(g) and C8H18(1) re...The enthalpy of neutralization of a strong monobasic acid by a strong monoacidic base is –13,700 cal. A certain monobasi...Calculate the standard free energy change for the ionization HF(aq) → H+ (aq) + F– (aq) from the following data.HF(aq) →...Given that :-CH4(g) + 300 kJ → C(g) + 4H(g)C2H6(g) + 620 kJ → 2C(g) + 6H(g)The value of C - C bond energy is...The standard molar enthalpies of formation of cyclohexane (l) and benzene(l) at 298 K are −156 and +49 kJ/ mol, respecti...