Mole ConceptHard
Question
The density of a liquid is 1.2 g/mL. There are 35 drops in 2 mL. The number of molecules in 1 drop is : (molecular weight of liquid = 70)
Options
A.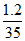 NA
NA
B.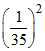 NA
NA
C.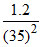 NA
NA
D.1.2 NA
Solution
Volume of one drop = 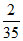 mL
mL
Number of moles in one drop =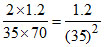
Number of molecules is one drop =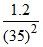 × NA
× NA
Number of moles in one drop =
Number of molecules is one drop =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In Duma′s method of estimation of nitrogen 0.35 g of an organic compound gave 55 mL of nitrogen collected at 300 K...The temperature at which molarity of pure water is equal to its molality is :...Accumulation of which of the following molecules in the muscles occurs as a result of vigorous exercise ?...Indentify the likely organisms (a), (b) (c) and (d) in the food web shown below...The vapour density of gas A is four times that of B. If molecular mass of B is M, then the molecular mass of A is :-...