s-Block ElementsHard
Question
In order to accomplish the following conversion, what reagent and conditions would be required?
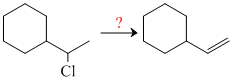

Options
A.Cold sodium hydroxide
B.Hot conc. sodium hydroxide
C.Potassium t-butoxide and heat
D.Hot water
More s-Block Elements Questions
Based on lattice energy and other considerations which one of the following alkali metal chlorides is expected to have t...Which of the following disproportionate(s) on heating with sodium hydroxide ?...The pair of amphoteric hydroxides is :...Select the correct statement with respect to alkali metals....A metal salt solution forms a yellow precipitate with potassium chromate in acetic acid, a white precipitate with dil H2...