s-Block ElementsHard
Question
Which of the following disproportionate(s) on heating with sodium hydroxide ?
Options
A.P4
B.S8
C.Cl2
D.B.
Solution
(A) P4 + 3 NaOH + 3 H2O → PH3 + 3 NaH2PO2
(B) 4S + 6 NaOH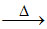 Na2S2O3 + 2 Na2S + 3 H2O
Na2S2O3 + 2 Na2S + 3 H2O
(C) 3 Cl2 + 6 NaOH → 5 NaCl + NaClO3 + 3 H2O
(D) 2B + 6 NaOH → 2 Na3BO3 + 3 H2
(B) 4S + 6 NaOH
(C) 3 Cl2 + 6 NaOH → 5 NaCl + NaClO3 + 3 H2O
(D) 2B + 6 NaOH → 2 Na3BO3 + 3 H2
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which of the following product is obtained as insoluble, when ammonical solution of brine is saturated with carbon dioxi...Which is correct statement...Which of the alkaline earth metal halides given below is essentially covalent in nature ?...Among the following components of cement which is present in highest amount ?...Calcium cyanamide on reaction with steam under pressure gives ammonia and -------....