SolutionHard
Question
Ratio of 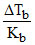 of 10g AB2 and 14g A2B per 100g of solvent in their respective, solution (AB2 and A2B both are non-electrolytes) is 1 mol/kg in both cases. Hence, atomic wt. of A and B are respectively:-
of 10g AB2 and 14g A2B per 100g of solvent in their respective, solution (AB2 and A2B both are non-electrolytes) is 1 mol/kg in both cases. Hence, atomic wt. of A and B are respectively:-
Options
A.100, 40
B.60, 20
C.20, 60
D.None of these
Solution
ᐃTb = Kb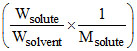 × 1000
× 1000
1 =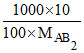 ⇒ MAB2 = 100
⇒ MAB2 = 100
similarly MAB2 = 140
100 = MA + 2.MB
and 140 = 2.MA + MB ;
MA = 60 and MB = 20
1 =
similarly MAB2 = 140
100 = MA + 2.MB
and 140 = 2.MA + MB ;
MA = 60 and MB = 20
Create a free account to view solution
View Solution FreeMore Solution Questions
If molality of the dilute solutions is doubled, the value of molal depression constant (Kf) will be :-...Among the following the surfactant that will form micelles in aqueous solution at the lowest molar concentration at ambi...The solubility of solid silver chromate, Ag2CrO4 is determined in three solvents. (Ksp = 9 × 10-12)(i) Pure water (...In mixture A and B component show - ve deviation as...For a dilute solution containing 2.5 g of a non-volatile non-electrolyte solute in 100 g of water, the elevation in boil...