Chemical EquilibriumHard
Question
Consider the following reversible reactions (at 298 K) :
(a) N2O4 ⇋ 2NO2
(b) 2SO2 + O2 ⇋ 2SO3
(c) 2HI ⇋ H2 + I2
(d) X + Y ⇋ 4Z
Highest and lowest value of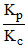 will be shown by the equilibrium
will be shown by the equilibrium
(a) N2O4 ⇋ 2NO2
(b) 2SO2 + O2 ⇋ 2SO3
(c) 2HI ⇋ H2 + I2
(d) X + Y ⇋ 4Z
Highest and lowest value of
Options
A.b, d
B.a, c
C.a, b
D.b, c
More Chemical Equilibrium Questions
For the reaction H2(g) + I2(g) $\rightleftharpoons$2HI(g), the rate law expression is...For the reaction 2NOCl(g)$\rightleftharpoons$ 2NO(g) + Cl2(g), ΔHo = 18 kcal and ΔSo = 30 cal/K at 300 K. The equilibriu...Two gases A and B, one being the dimer of the other, are at equilibrium. Decreasing of pressure at constant temperature ...For the reaction CaCO3(s) $\rightleftharpoons$CaO(s) + CO2(g), the value of KP is...For a gaseous equilibrium 2A(g) $\rightleftharpoons$2B(g) + C(g), KP has a value 1.8 at 700 K. The value of KC for the e...