p-Block elementsHard
Question
Which of the following is/are true for oxygen.
Options
A.KMnO4(s) on strong heating gives oxygen gas
B.Oxygen mixed with helium is used for artificial respiration.
C.It has two unpaired electrons in bonding p molecular orbitals.
D.Brins process is used as industrial method for the preparation of oxygen gas.
Solution
(A) 2KMnO4 → K2MnO4 + MnO2 + O2
(B) He is insoluble in blood at higher pressure.
(C) O2 has two unpaired electrons in antibonding p molecular orbitals.
(D) 2BaO + O2 (air)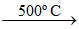 2BaO2 ; BaO2
2BaO2 ; BaO2 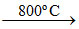 2BaO + O2
2BaO + O2
(B) He is insoluble in blood at higher pressure.
(C) O2 has two unpaired electrons in antibonding p molecular orbitals.
(D) 2BaO + O2 (air)
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following metals does not dissolve in concentrated HNO3?...The strongest acid amongst the following is :...With respect to graphite and diamond, which of the statement(s) given below is (are) correct?...Cold solution of barium nitrite on mixing with sulphuric acid produces :...The hydroxide of which metal ion is soluble in excess of sodium hydroxide solution :...