ThermodynamicsHard
Question
A gas takes part in three thermal processes in which it is heated from the same initial state to the same final temperature. The process are shown on the P - V diagram by straight lines 1-2, 1-3 and 1-4. Q1, Q2 and Q3 are amount of heat supplied to the gas in the process 1 → 2, 1 → 3, 1 → 4, respectively.
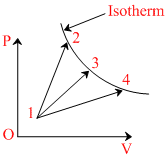

Options
A.Q3 > Q2
B.Q1 < Q2
C.Q3 > Q1
D.Q3 > Q2 > Q1τ
More Thermodynamics Questions
If cp and cv denotes the specific heats (per unit mass of an ideal gas of molecular weight M, then...An ideal gas is taken round the cycle ABCA. In the cycle the amount of work done involved is: -...The relative humidity on a day, when partial pressure of water vapour is 0.02 × 105 pa at 12oC is (take vapoure pre...Which of the following statements is correct for any thermodynamic system?...The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K theroot mean square velocity of the gas mo...