Carboxylic Acid and Acid DerivativesHard
Question
Identify the binary mixture(s) that can be separated into individual compounds, by differential extraction, as shown in the given scheme.
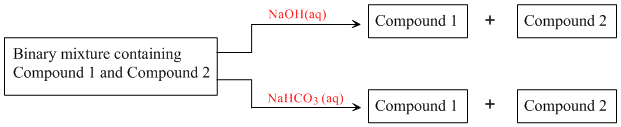

Options
A.C6H5OH and C6H5COOH
B.C6H5COOH and C6H5CH2OH
C.C6H5CH2OH and C6H5OH
D.C6H5CH2OH and C6H5CH2COOH
Solution
(A) Both are soluble in NaOH, hence inseparable.
(B) Only benzoic acid (C6H5COOH) is soluble in NaOH and NaHCO3, while benzyl alcohol (C6H5CH2OH) is not. Hence, separable.
(C) Although NaOH can enable separation between benzyl alcohol (C6H5CH2OH) and phenol (C6H5OH) as only the later is soluble in NaOH. However, in NaHCO3, both are insoluble. Hence, inseparable.
(D) α - phenyl acetic acid (C6H5CH2COOH) is soluble in NaOH and NaHCO3. While benzyl alcohol (C6H5CH2OH) is not. Hence, separable.
(B) Only benzoic acid (C6H5COOH) is soluble in NaOH and NaHCO3, while benzyl alcohol (C6H5CH2OH) is not. Hence, separable.
(C) Although NaOH can enable separation between benzyl alcohol (C6H5CH2OH) and phenol (C6H5OH) as only the later is soluble in NaOH. However, in NaHCO3, both are insoluble. Hence, inseparable.
(D) α - phenyl acetic acid (C6H5CH2COOH) is soluble in NaOH and NaHCO3. While benzyl alcohol (C6H5CH2OH) is not. Hence, separable.
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Why − OH group in ethyl alcohol is neutral, while it is acidic in acetic acid -...Hydrolysis of which one of the following compound is easier -...A compound with molecular formula C4H10O4 on acylation with acetic anhydride gives a compound with molecular formula C12...Carboxylic acid have higher boiling points than aldehydes, ket ones an d even alcoh ols of comparable molecular mass. It...Which of the following is correct order of acidic strength among :-(i) CO2 (ii) SiO2 (iii) SO3 (iv) N2O5...