NEET | 2018Carboxylic Acid and Acid DerivativesHard
Question
Carboxylic acid have higher boiling points than aldehydes, ket ones an d even alcoh ols of comparable molecular mass. It is due to their
Options
A.
formation of intramolecular H-bonding
B.
formation of carboxylate ion
C.
more extensive association of carboxylic acid via van der Waals force of attraction
D.
formation of intermolecular H-bonding.
Solution
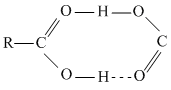
Carboxylic acid has higher boiling point than aldehyde, ketone and even alcohols of comparable molecular mass. This is due to more extensive association through intermolecular H-bonding.
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
In the given reaction : [X] will be :...A reaction of ethyl amine & acetic anhydride leads to the formation of :...Hydrolysis of which one of the following compound is easier -...Benzoic acid on treatment with hydrazoic acid in the presence of concentrated sulphuric acid gives:...The compound obtained by the reaction between primary amine and aldehyde is:...