JEE Advanced | 2013Carboxylic Acid and Acid DerivativesHard
Question
The compound that does NOT liberate CO2, on treatment with aqueous sodium bicarbonate solution,is -
Options
A.Benzoic acid
B.Benzenesulphonic acid
C.Salicylic acid
D.Carbolic acid (phenol)
Solution
Acid base reaction
HA + NaHCO3 ⇋ H2CO3 + Na+A- (1)
(acid) (aq.)
(1) Above acid base equilibrium favours in forward if acid (HA) is stronger then H2CO3
(2) Benzoic acid (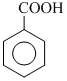 ) , benzene sulphonic acid (
) , benzene sulphonic acid (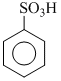 ) salicylic acid (
) salicylic acid (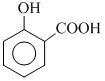 ) are stronger acid than H2CO3. So above equilibrium favours in forward direction and unstable H2CO3 formed librates CO2 gas by following reaction
) are stronger acid than H2CO3. So above equilibrium favours in forward direction and unstable H2CO3 formed librates CO2 gas by following reaction
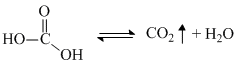
(3) Phenol is less acidic than H2CO3 so it cannot protonate to NaHCO3 and CO2 gas does not evolve.
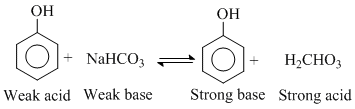
Ans. phenol doesn′t evolve CO2 with NaHCO3
HA + NaHCO3 ⇋ H2CO3 + Na+A- (1)
(acid) (aq.)
(1) Above acid base equilibrium favours in forward if acid (HA) is stronger then H2CO3
(2) Benzoic acid (
 ) , benzene sulphonic acid (
) , benzene sulphonic acid ( ) salicylic acid (
) salicylic acid ( ) are stronger acid than H2CO3. So above equilibrium favours in forward direction and unstable H2CO3 formed librates CO2 gas by following reaction
) are stronger acid than H2CO3. So above equilibrium favours in forward direction and unstable H2CO3 formed librates CO2 gas by following reaction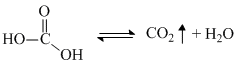
(3) Phenol is less acidic than H2CO3 so it cannot protonate to NaHCO3 and CO2 gas does not evolve.

Ans. phenol doesn′t evolve CO2 with NaHCO3
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
The end product in the reaction CH3CONH2 B C D E is -...Which of the following pairs produces deutronitric acid :-Which of the following pairs produces deutronitric acid :-...Acetic anhydride and ammonia gives the product :...Which one of the following compounds is least reactive with water?...Which one is the correct order of acidity ?...