Carboxylic Acid and Acid DerivativesHard
Question
Which of the following undergoes haloform reaction ?
(i) CH3CH2 COCH2Cl (ii) C6H5COCH3
(iii) C6H5COCHCl2 (iv) CH3CH2COCCl3
(i) CH3CH2 COCH2Cl (ii) C6H5COCH3
(iii) C6H5COCHCl2 (iv) CH3CH2COCCl3
Options
A.only (ii)
B.(ii) and (iv)
C.(i), (ii) and (iv)
D.all the four
Solution
If we observe the haloform reaction carefully, we see that - COCH3 group is first halogented to the trihalo- COCX3 through monohalogeno ang dihalogeno compound. It is the - COCX3 patr which then undergoes nuclhilic adition. the produccy easily loses-CX3 since it is a very good leaving group.
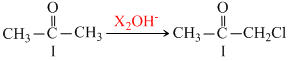
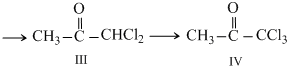
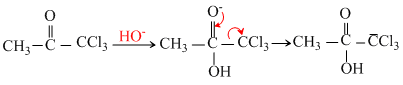
Thus all compounds (I to IV)are ultimatelyconverted to CHCl3(chloroform).
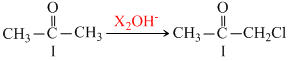
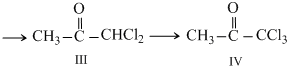

Thus all compounds (I to IV)are ultimatelyconverted to CHCl3(chloroform).
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Which one of the following acids undergoes decarboxylation on strong heating:...Extra pure N2 can be obtained by heating...Which acid can be oxidised by Fehling solution :...Acetamide is treated separately with the following reagnts. Which one of these would give methyl amine ?...In the given reaction :CH3 - COOH [X][X] will be:...