Alkyl Halides and Aryl HalidesHard
Question
CH3OC2H5 and (CH3)3 C - OCH3 are treated with hydriodic acid. the fragments obtained after reactions are
Options
A.CH3I + HOC2H5; (CH3)3CI + HOCH3
B.CH3OH + C2H5I;(CH3)3CI + HOCH3
C.CH3OH + C2H5I;(CH3)3COH + CH3I
D.CH3I+ HOC2H5; CH3I(CH3)3COH.
Solution
In case of unsymmetrical ethers,the site of cleage depends on the nature of alkyl group.
e.g., CH3 - O - C2H5 + HI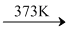 CH3I + C2H5OH
CH3I + C2H5OH
The alkyl haide is formed from the smaller alkyl group.
However, in case of teritiary alkyl ether following reaction occurs.
CH3- O -(CH3)3 + HI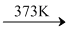 (CH3)3 C - I + CH3 OH
(CH3)3 C - I + CH3 OH
ter-butyl methyl ether
Tnhe alkyl halide is formed from the tertiary alkyl group and the cleavage of such ethers occurs by SN1mechanism as the product is controlled by the formation of more stable intermedied ether.
e.g., CH3 - O - C2H5 + HI
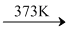 CH3I + C2H5OH
CH3I + C2H5OHThe alkyl haide is formed from the smaller alkyl group.
However, in case of teritiary alkyl ether following reaction occurs.
CH3- O -(CH3)3 + HI
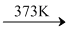 (CH3)3 C - I + CH3 OH
(CH3)3 C - I + CH3 OHter-butyl methyl ether
Tnhe alkyl halide is formed from the tertiary alkyl group and the cleavage of such ethers occurs by SN1mechanism as the product is controlled by the formation of more stable intermedied ether.
Create a free account to view solution
View Solution FreeMore Alkyl Halides and Aryl Halides Questions
Which of the following statement is correct -...The reaction C2H2OH + SOCl2 C2H5Cl + SO2 + HCl is known as...which one of the following is primary halide?...Which of the following compounds is most rapidly hydrolysed by SN1 mechanism....Which of the following compounds will undergo racemisation when solution of KOH hydrolyzes? (i) (ii) CH3CH2CH2Cl (iii) (...