Alkyl Halides and Aryl HalidesHard
Question
Intermolecular hydrogen bonding is strongest in :
Options
A.Methylamine
B.Phenol
C.Formadehyde
D.Methanol
Solution
Hydrogen bonding is formed in compounds in which H is attached to highly electronegative element like F, O and N.
(i) In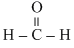 , O is not having H atom so it shows very little H-bonding.
, O is not having H atom so it shows very little H-bonding.
(ii) N is less electronegative than O, so H-bond formed by amines will be weak than that by alcohols.
(i) In
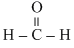 , O is not having H atom so it shows very little H-bonding.
, O is not having H atom so it shows very little H-bonding.(ii) N is less electronegative than O, so H-bond formed by amines will be weak than that by alcohols.
Create a free account to view solution
View Solution FreeMore Alkyl Halides and Aryl Halides Questions
The order of solubility of lithium halides in non polar solar solvents follows the order :...Which of the following on alkaline hydrolysis gives ethanal -...Arrange the following compounds in order of decreasing rate of hydrolysis for SN1 reaction:(I) (II)(III) (IV)...The reactivity of 2-bromo-2-methylbutane (I), 1-bromopentane (II) and 2bromopentane (III) towards SN2 displacement is su...The reaction of silver carboxylates with bromine dissolved in carbon tetrachloride is called -...