Alkyl Halides and Aryl HalidesHard
Question
0.5g of sample of bleaching powder was suspended in water and an exess KI added. Onacidifying with dil. H2SO4, I2 was liberated which required 50 cc of N/10 hypo solution. the percentage of Cl2 in bleaching powder is :
Options
A.35.5%
B.45.5%
C.25.5%
D.39.5%
Solution
The concerned chemical reactions are
CaOCl2 + 2CH3COOH → (CH3COO)2Ca + H2O + Cl
2Kl + Cl2 → 2KCl + I2
I2 + 2Na2S2O3 → Na2S4O6 + 2Nal
Thus meq of hypo = meq of I2 = meq of Cl2
≡ 50 ×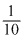 = 5
= 5
We know that meq of Cl2 =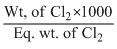
5 =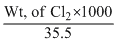
Wt.of Cl2 =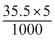 = 0.1775g
= 0.1775g
% of Cl2 =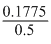 × 100 = 35.5%
× 100 = 35.5%
CaOCl2 + 2CH3COOH → (CH3COO)2Ca + H2O + Cl
2Kl + Cl2 → 2KCl + I2
I2 + 2Na2S2O3 → Na2S4O6 + 2Nal
Thus meq of hypo = meq of I2 = meq of Cl2
≡ 50 ×
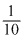 = 5
= 5We know that meq of Cl2 =
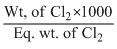
5 =
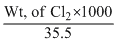
Wt.of Cl2 =
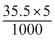 = 0.1775g
= 0.1775g% of Cl2 =
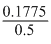 × 100 = 35.5%
× 100 = 35.5%Create a free account to view solution
View Solution Free